Operating Conditions of MFC
So far, performances of laboratory MFCs are still much lower than the ideal performance. There may be several possible reasons. Power generation of an MFC is affected by many factors including microbe type, fuel biomass type and concentration, ionic strength, pH, temperature, and reactor configuration. Effects of reactor configuration and types of microbe used in the MFC have been addressed in Sections 2 and 3. With a given MFC system, the follow-ing operating parameters can be regulated to decrease the polarizations in order to enhance the performance of an MFC.
Effect of electrode materials
Using better performing electrode materials can improve the performance of an MFC because different anode materials result in different activation polarization losses. Pt and Pt black electrodes are superior to graphite, graphite felt and carbon-cloth electrodes for both anode and cathode constructions, but their costs are much higher.Schroder et al. (2003) reported that a current of 2-4 mA could be achieved with platinu-mized carbon-cloth anode in an agitated anaerobic cul-ture of E. coliusing a standard glucose medium at 0.55 mmol/L, while no microbially facilitated current flow is observed with the unmodified carbon-cloth with the same operating conditions. Pt also has a higher catalytic activity with regard to oxygen than graphite materials. MFCs with Pt or Pt-coated cathodes yielded higher power densities than those with graphite or graphite felt cathodes.
pH buffer and electrolyte
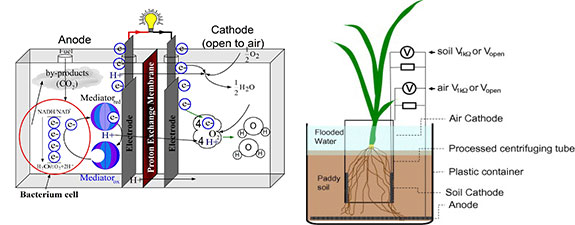
If no buffer solution is used in a working MFC, there will be an obvious pH difference between the anodic and cathodic chambers, though theoretically there will be no pH shift when the reaction rate of protons, electrons and oxygen at the cathode equals the production rate of protons at the anode. The PEM causes transport barrier to the cross membrane diffusion of the protons, and proton transport through the membrane is slower than its production rate in the anode and its consumption rate in the cathode chambers at initial stage of MFC operation thus brings a pH difference. However, the pH difference increases the driving force of the proton diffusion from the anode to the cathode chamber and finally a dynamic equilibrium forms. Some protons generated with the biodegradation of the organic substrate transferred to the cathodic chamber are able to react with the dissolved oxygen while some protons are accumulated in the anodic chamber when they do not transfer across the PEM or salt bridge quickly enough to the cathodic chamber. Gil et al. (2003) detected a pH difference of 4.1 (9.5 at cathode and 5.4 in anode) after 5-hour operations with an initial pH of 7 without buffering. With the addition of a phosphate buffer (pH 7.0), pH shifts at the cathode and anode were both less than 0.5 unit and the current output was increased about 1 to 2 folds. It was possible that the buffer compensated the slow proton transport rate and improved the proton availability for the cathodic reaction. Jang et al. (2004) supplied an HCl solution to the cathode and found that the current output increased by about one fold. This again suggests that the proton availability to the cathode is a limiting factor in electricity generation. Increasing ionic strength by adding NaCl to MFCs also improved the power output, possibly due to the fact that NaCl enhanced the conductivity of both the anolyte and the catholyte.
Proton Exchange System
Proton exchange system can affect an MFC system's internal resistance and concentration polarization loss and they in turn influence the power output of the MFC. Nafion (DuPont, Wilmington, Delaware) is most popular because of its highly selective permeability of protons. Despite attempts by researchers to look for less expensive and more durable substitutes, Nafion is still the best choice. However, side effect of other cations transport is unavoidable during the MFC operation even with Nafion. In a batch accumulative system, for example, transportation of cation species other than protons by Nafion dominates the charge balance between the anodic and cathodic chambers because concentrations of Na+,K+,NH4+,Ca2+,Mg2+ are much higher than the proton concentrations in the anolyte and catholyte. In this sense, Nafion as well as other PEMs used in the MFCs are not a necessarily proton specific membranes but actually cation specific membranes.
The ratio of PEM surface area to system volume is important for the power output. The PEMsurface area has a large impact on maximum power output if the power output is below a critical threshold. The MFC internal resistance decreaseswith the increase of PEMsurface area over a relatively large range.
Operating conditions in the anodic chamber
Fuel type, concentration and feed rate are important factors that impact the performance of an MFC. With a given microbe or microbial consortium, power density varies greatly using different fuels. Table shows the performances of some MFCs operated using different microbes and fuels. Many systems have shown that electricity generation is dependent on fuel concentration both in batch and continuous-flow modeMFCs. Usually a higher fuel concentration yields a higher power output in a wide concentration range. Park and Zeikus (2002) reported that a higher current level was achieved with lactate (fuel) concentration increased until it was in excess at 200 mM in a single-compartment MFC inoculated with S. putrefaciens. Moon et al. (2006) investigated the effects of fuel concentration on the performance of an MFC. Their study also showed that the power density was increased with the increase in fuel concentration. Gil et al. (2003) found that the current increased with a wastewater concentration up to 50 mg/L in their MFC. Interestingly, the electricity generation in an MFC often peaks at a relatively low level of feed rate before heading downward. This may be because a high feed rate promoted the growth of fermentative bacteria faster than those of the electrochemically active bacteria in a mixed culture. However, if microbes are growing around the electrodes as biofilms, the increased feed rate is unlikely to affect the flora. One possible reason is that the high feed rate brings in other alternate electron acceptors competing with the anode to lower the output.
Operating conditions in the cathodic chamber
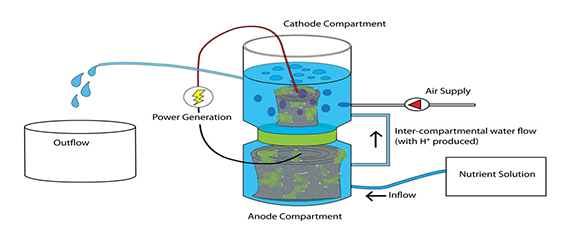
Oxygen is the most commonly used electron acceptor in MFCs for the cathodic reaction. Power output of an MFC strongly depends on the concentration level of electron acceptors. Several studies indicated that DO was a major limiting factor when it remained below the air-saturated level. Surprisingly, a catholyte sparged with pure oxygen that gave 38 mg/L DO did not further increase the power output compared to that of the air-saturated water (at 7.9 mg/L DO). Rate of oxygen diffusion toward the anode chamber goes upwith the DO concentration. Thus, part of the substrate is consumed directly by the oxygen instead of transferring the electrons though the electrode and the circuit. Power output is much greater using ferricyanide as the electron acceptor in the cathodic chamber. So far, reported cases with very high power outputs such as 7200 mW/m2,4310mW/m 2 and 3600 mW/m 2 all used ferricyanide in the cathodic chamber, while less than 1000 mW/m 2 was reported in studies using DO regardless of the electrode material. This is likely due to the greatermass transfer rate and lower activation energy for the cathodic reaction offered by ferricyanide . Using hydrogen peroxide solution as the final electron acceptor in the cathodic chamber increased power output and current density according to Tartakovsky and Guiot (2006).As a consequence, aeration is no longer needed for single-compartment MFCs with a cathode that is directly exposed to air. Rhoads et al. (2005) measured the cathodic polarization curves for oxygen and manganese and found
Surely changing operating conditions can improve the power output level of the MFCs. However, it is not a revolutionary method to upgrade the MFCs from low power system to a applicable energy source at the very present. The bottleneck lies in the low rate of metabolism of the microbes in the MFCs. Even at their fastest growth rate microbes are relatively slow transformers. The biotransformation rate of substrates to electrons has a fixed ceiling which is inherently slow. Effort should be focused on how to break the inherent metabolic limitation of themicrobes for theMFC application. High temperature can accelerate nearly all kinds of reactions including chemical and biological ones. Use of thermophilic species might benefit for improving rates of electron production, however, to the best of our knowledge, no such investigation is reported in the literature. Therefore this is probably another scope of improvement for the MFC technology fromthe laboratory research to a real applicable energy source.